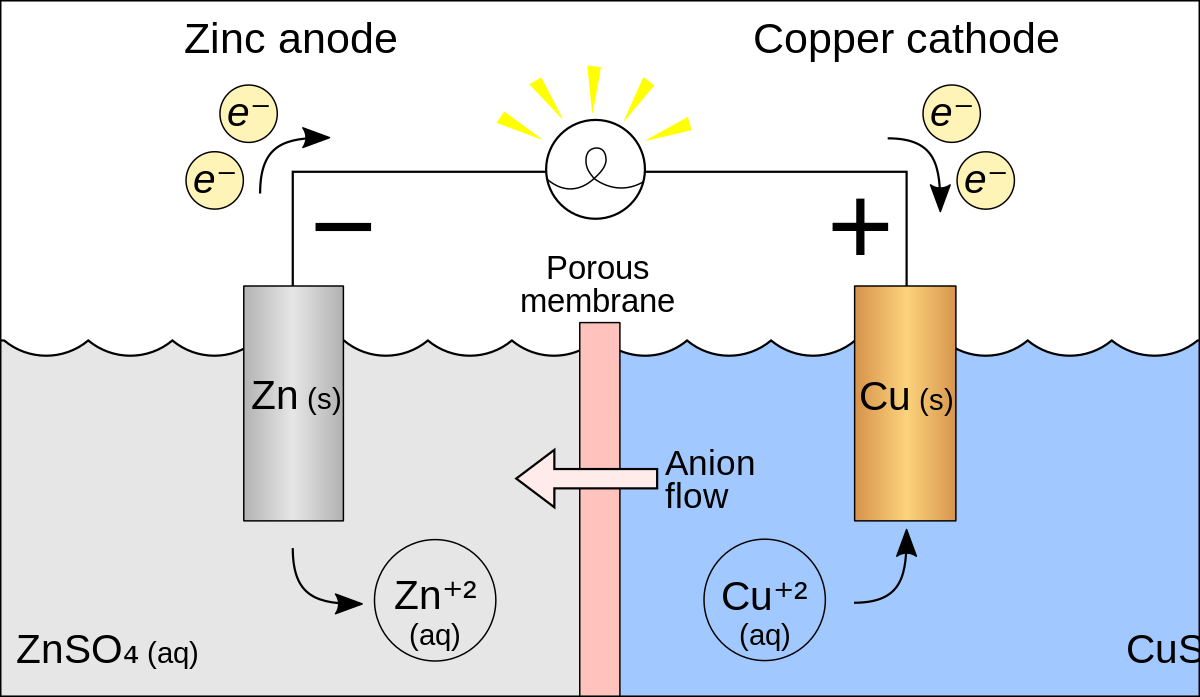
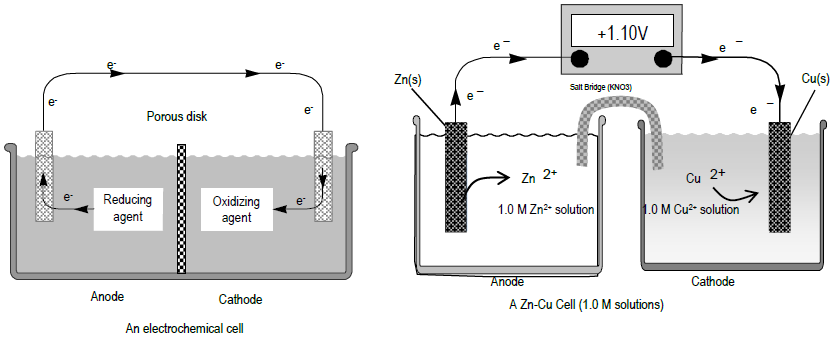
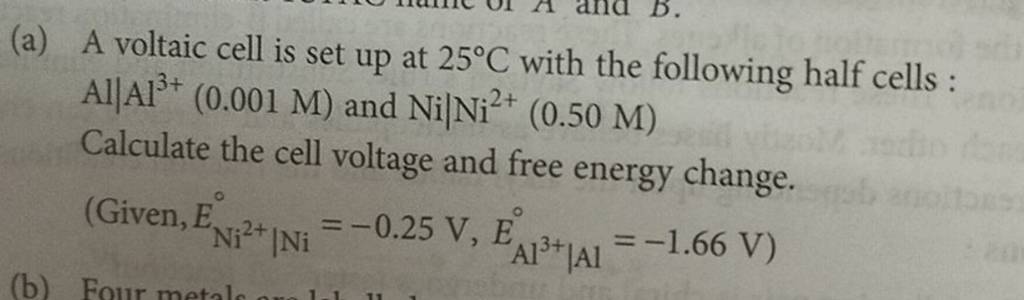A voltaic cell in set up at 25^oC with the following half cells: Al/Al^3 + (0.001 M) and Ni/Ni^2 + (0.50 M) Write an equation for the reaction that occurs when the

SOLVED: A voltaic cell is set up at 25 C with the half cells Sn+2 (0.050 M) and Ag (0.0250 M). Sketch the voltaic cell. Be sure t0 label the anode, cathode
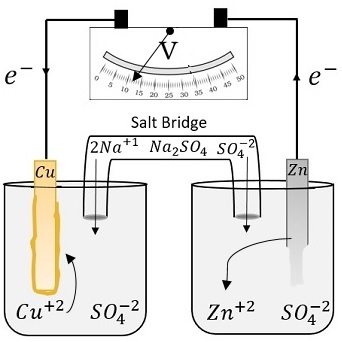
A voltaic cell is set up 25° C with the following half-cells: - Sarthaks eConnect | Largest Online Education Community

✓ Solved: A silver concentration cell is set up at 25^°C as shown below: The AgCl(s) is in excess in...
A voltaic cell is set up 25° C with the following half-cells: - Sarthaks eConnect | Largest Online Education Community
A voltaic cell is set up at 25°C with the following half cells. Al | Al^3+ (0.0010 M) and Ni^2+ (0.50 M) | Ni - Sarthaks eConnect | Largest Online Education Community

taic cell is up 25 °C with the following half cells, 9. 20. A voltaic cell is ur A bovlofti eroinsta A13+ and NiNi2+ (0.001 M) (0.5 M) EX13+ / Al = -

taic cell is up 25 °C with the following half cells, 9. 20. A voltaic cell is ur A bovlofti eroinsta A13+ and NiNi2+ (0.001 M) (0.5 M) EX13+ / Al = -

A voltaic cell in up 25oC with the following half cells:Al/Al3+(0.001M) and Ni/Ni2+(0.50M)Write an equation the reaction that occurs when the cell generates an electric current and determine the cell potential. ENi2+/Ni=−0.25V




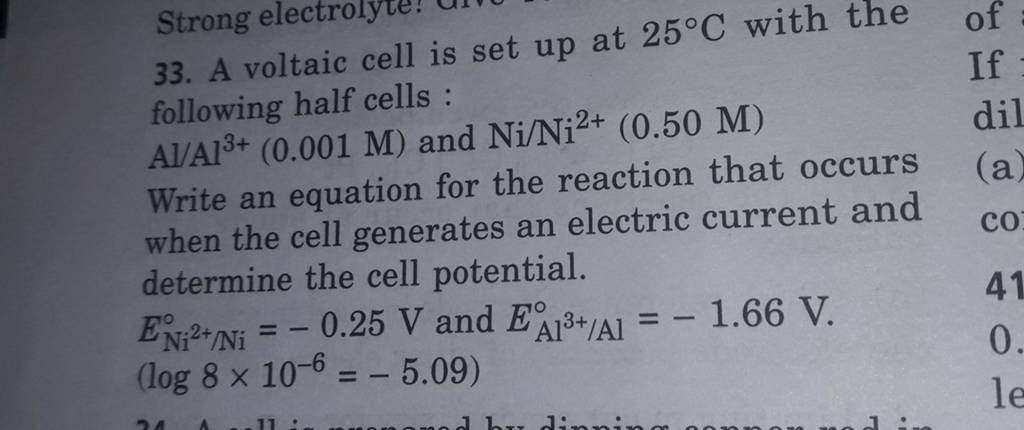


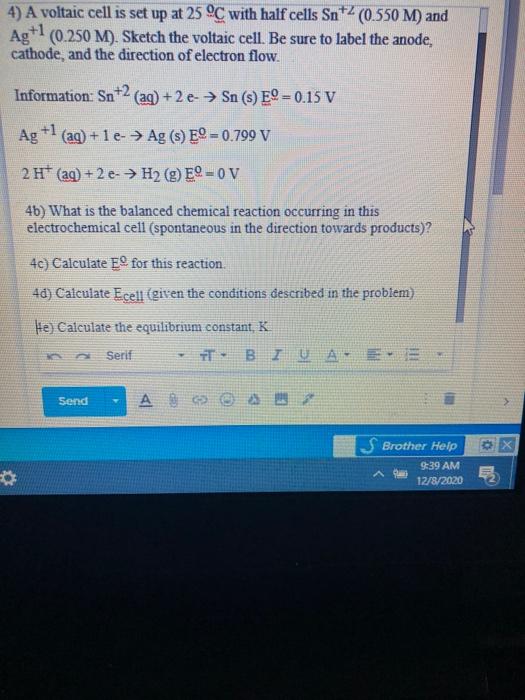



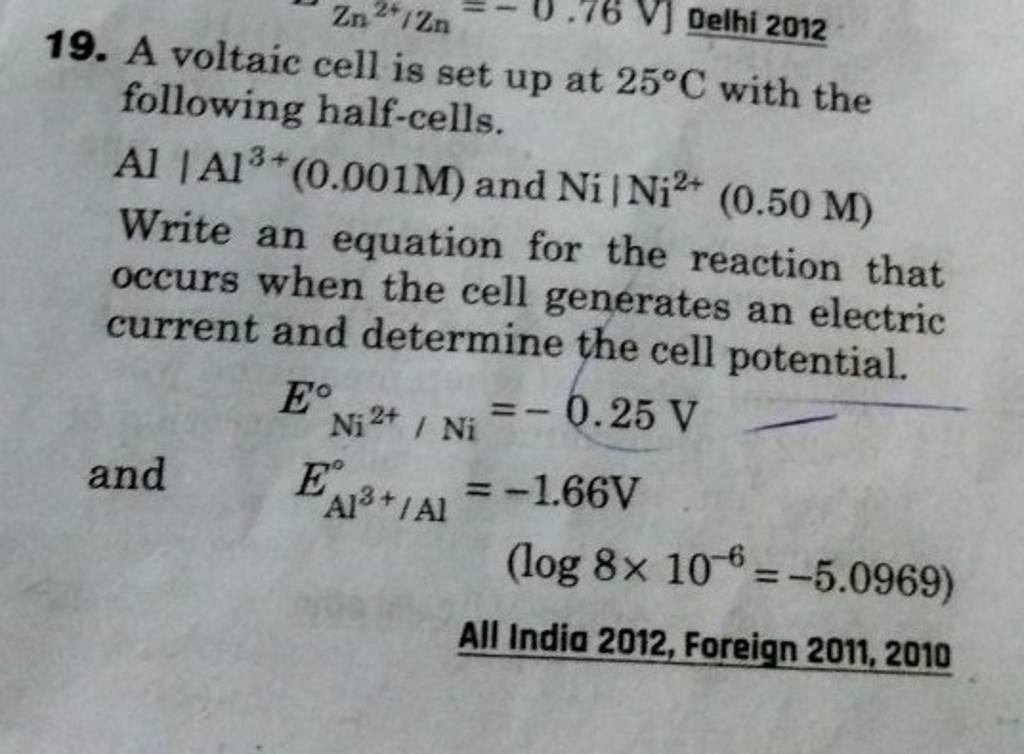
![Kannada] A voltic cell is set up at 25^@C with the half cells Ag^(+) Kannada] A voltic cell is set up at 25^@C with the half cells Ag^(+)](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/7681973.webp)